INTRODUCTION:
Two experiments are actually combined into one through the concept
of rings or cyclic structures, either in the monomer or the polymer
formed. Seems a little tenuous, but these procedures are short and it
does give you a different perspective on things. Background information
is given on the general concepts of ring-opening and ring-forming
polymerizations that includes industrial examples and mechanisms. The
polymer synthesis for each type is combined with characterization by IR
spectroscopy and dilute solution viscosity. The latter illustrates the
polyelectrolyte effect for the charged ammonium polymer in water.
Polyamines, polyamine salts and polyamides are used commercially in a
wide range of applications. These include sizing for paper and
textiles; recovery and recycle of trace metal contaminates from chemical
plants; and flocculation of particulate matter for water clarification.
These polymers are strong complexing and chelating agents for metal
salts, and such complexes have been used for a variety of catalytic
applications. In addition, many of the polyamine compounds are basic
catalysts in their own right or in conjunction with organic comonomers,
and have been used to make a variety of synthetic chemicals.
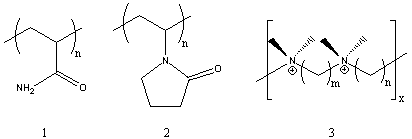
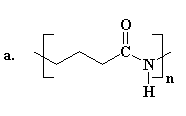
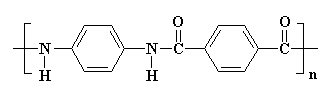
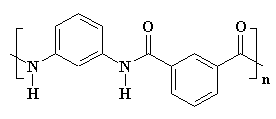
Several of the presently available commercial polymers containing
amide and ammonium functionality are given in Figure 1. These included
vinyl addition polymers from acrylamide (1) and N-vinylpyrrolidone (2),
and step-growth polymers containing quaternary ammonium groups
(polyionenes, 3) obtained from polycondensation of diamines and
bishalides.
Figure 1. Commercial Amide and Ammonium Polymers.
In this experiment, we examine two major types of polymerization
processes involving heterocyclic monomers or repeat units. Polymer
synthesis involves ring-opening polymerization to yield a polyamide from
an oxazoline, and cyclopolymerization of diallylamine derivative to
yield a polymer containing pyrrolidine units.
Ring-opening polymerization mechanisms
There are several commercially important polymers which are
synthesized via ring-opening polymerization. Examples summarized in
Figure 2 include such common polymers as polyoxyethylene (POE, 4),
poly(butylene oxide) (PBO, 5), nylon 6 (6), and poly(ethyleneimine)
(PEI, 7). This last polymer is obtained by a non-selective process
which can involve attack on the ethyleneimine monomer by either
chain-ends or internal secondary amines of the growing polymer. These
competing reactions lead to a highly branched polymer structure which
contains primary, secondary and tertiary amine units.1
Figure 2. Common Ring-Opening Polymerizations. 
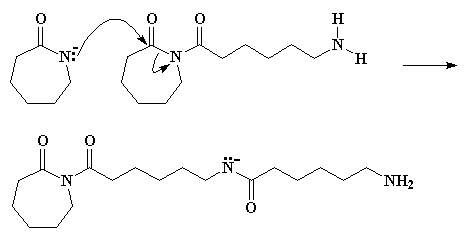
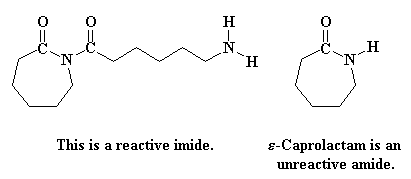
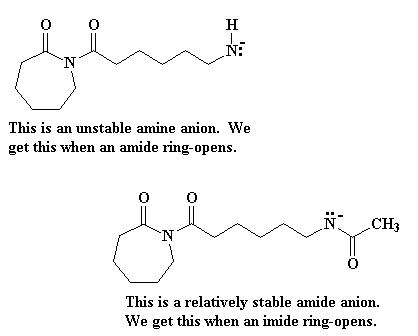
Several years ago, a novel synthesis of completely linear PEI was developed.2
The method utilized a ring-opening polymerization also, but of a
5-membered heterocycle that resulted in formation of a substituted amide
rather than the free amine obtained from ethyleneimine. Figure 3
summarizes the initiation and propagation steps for this polymerization 3 as well as the hydrolysis reaction and the final polymer structure.
Figure 3. Ring-Opening Polymerization Mechanism for 2-Substituted Oxazolines
and Subsequent Polymer Hydrolysis; the Circled "P" Represents Polymer
Chain with the Indicated Active Chain End. 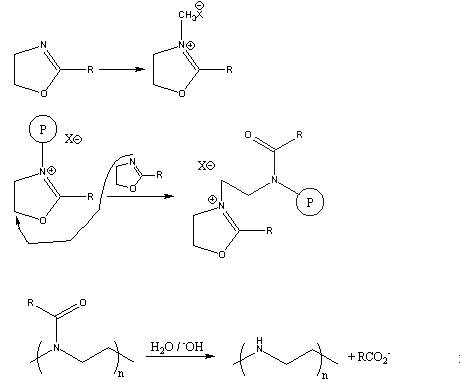
This method gives linear PEI (8) by a two-step process. In
addition, the intermediate polymers containing amide functionality have
become important in their own right.4 Many of these polymers
are being examined for various unique applications involving a
combination of properties. Many are soluble both in water and in a wide
range of organic solvents. The amide functionality provides multiple
sites for complexation and chelation of a variety of metal salts. The
controlled spacing of the pendent amide derivatives along the 3-atom
repeat unit in the backbone provides a novel alternative to the normal
2-atom backbone obtained with vinyl polymerization (see for example
poly-(N-vinylpyrrolidone), 2). Finally, partial hydrolysis can give
polymers containing both amide and amine or ammonium groups which can
interact with substrates, together or in a sequential fashion.
The most common oxazoline derivative available today is the 2-ethyl
compound. In this experiment, the monomer is polymerized using a
cationic initiator to give high molecular weight polymer which is
characterized by both IR and solubility behavior.
Cyclopolymerization
Cyclopolymerizations were first discovered by Professor George Butler in the late '50's.5
Since then, a wide variety of monomers have been found to undergo
cyclopolymerization. We concentrate here on a diallylamine derivative.
The cyclopolymerization process involves formation of a heterocyclic
ring during polymerization as illustrated in Figure 4. The monomer
shown, diallyldimethylammonium chloride, is one of the most widely used
commercial derivatives.
The cyclopolymerization mechanism involves two sequential propagation steps.6
Intermolecular attack of a propagating radical is immediately followed
by an intramolecular attack to form the heterocycle. Surprisingly,
this second step leads to the unstable primary radical through kinetic
rather than thermodynamic control, and is followed by immediate reaction
with another monomer molecule. Two possible side reactions can occur
in these polymerizations, involving crosslinking and chain transfer, but
they are not observed. In general, cyclopolymerization of
diallylammonium compounds proceeds cleanly to high molecular weight with
no crosslinking.
Figure 4. Free Radical Cyclopolymerization Mechanism of a
Diallylammonium Monomer 
Figure 5. Free Radical Cyclopolymerization Alternate Mechanism
of a Diallylammonium Monomer. 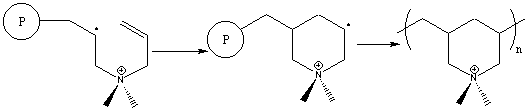
One of the earlier drawbacks in such polymerizations involved the
use of peroxide initiators. Extensive yellowing of the product polymer
and inefficient initiation lead to low yields and undesirable
properties. A recently reported improvement on these polymerizations
involves a new commercial initiator V-50 (2,2'-azobis(2-amidinopropane .
2HCl, 9). This water-soluble species cleanly forms carbon radicals
that initiate diallylammonium cyclopolymerization to high yield.7
In this experiment, the polymerizability of diallyldimethylammonium
chloride is examined. The polymer is purified by precipitation from
water and characterized by dilute solution viscosity.
EXPERIMENTAL:
Polymerization of 2-Ethyloxazoline
A clean, dry test tube is fitted with a rubber septum fastened on with wire. Approximately 2 ml of 2-ethyloxazoline8 is injected into the test tube which is then suspended in an oil bath preheated to 120oC. After a few minutes equilibration, the test tube is carefully removed and approximately 5ml of dimethylsulfate8
is injected. The test tube is put back in the oil bath. The solution
gradually becomes more viscous until it gels or solidifies (about 2 h).
The test tube is removed from the oil bath and allowed to cool. After
removing the septum, 5 ml of methylene chloride is added to dissolve the
mixture. This solution is then poured slowly into 50 ml of rapidly
stirring mixed hexanes. The solvent is carefully decanted from the
solid polymer which is washed again with more hexanes and finally
isolated by filtration. CAUTION: dimethylsulfate is toxic and should be handled only in small quantities with good ventilation.
Polymerization of Diallyldimethylammonium Chloride
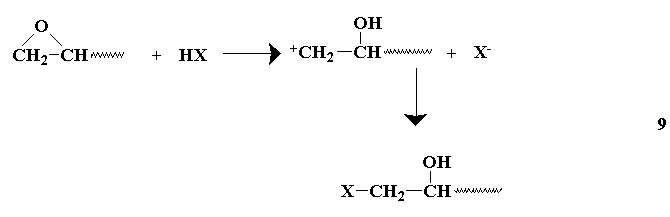
Commercial monomer is usually available at 65 wt-% solution in water.9
This is suitable for direct polymerization. Approximately 5 ml of
this solution is added to a test tube along with initiator 9 (V-509, about 0.05 g, ca. 1 mol-%). A septum cap is wired in place and the reaction mixture purged for 5-10 min with N2 through inlet and outlet needles in the septum. The test tube is placed in a preheated water or oil bath at 60-65oC. A small diameter syringe needle is left in the septum to relieve pressure from liberated N2
gas. Polymerization takes place rapidly to give a gelled or solid mass
within 1-2 h. The polymer is isolated by precipitation into 100 ml
ethanol stirring rapidly in a 250 ml beaker. Purification can be
carried out by reprecipitation from water into ethanol.
DISCUSSION:
The two synthetic procedures are straight-forward and can be carried
out with a minimum of special preparations and precautions. However,
dimethylsulfate is toxic. Only enough material for immediate use should
be used. The polymers are important commercially and represent less
well-known specialty chemicals. In addition, their synthesis introduces
the student to heterocyclic compounds in the context of polymer
formation.
Two synthetic extensions of the experiment are possible. One involves synthesis of a poly(diallylamine)7,
a polymer that is more difficult to purify and characterize.
(Diallylamine is also toxic). Alternatively, the oxazoline polymer can
by hydrolyzed in refluxing aqueous acid and neutralized to obtain the
free amine polymer. These two polymers can then be compared with the
other amide and amine polymers made in this experiment.
Polymer characterization involves qualitative evaluation of
solubility behavior, dilute solution viscosity, and IR spectroscopy.
Solubility should be evaluated for common organic solvents, acetic acid,
and aqueous acid and base solutions. The results can be compared with
other available polymers. The students must be made aware of the
importance of allowing sufficient time for dissolution and swelling to
take place (5-12 h). Unlike low molecular weight materials which
normally dissolve rapidly or not at all, polymers take appreciable time
to untangle and move away from the solid polymer mass. This effect
becomes more pronounced the higher the molecular weight of the polymer.
Dilute solution viscosity is one of the most common and useful
initial characterization techniques for polymers. At the very least, a
viscosity value of more than ca. 0.1 dL/g tells you that you do have a
polymer. More important, qualitative comparisons are possible for
polymers of the same composition; ie., increasing viscosity values
correlate directly with increasing molecular weight and polymer size.
Detailed procedures have been published previously for viscosity
determinations.
One very interesting aspect of the viscosity behavior of
poly(diallyldimethylammonium chloride) is the polyelectrolyte effect
that it shows.12 While well-behaved polymers show a linear
relationship with respect to concentration, polyelectrolytes usually
show higher reduced viscosity with decreasing concentration. This is
demonstrated in the upper plot of Figure 6 (viscosity with units of dL/g
plotted against concentration in g/dL). Addition of electrolytes
(NaCl) at relatively high concentrations ( > 0.5M) compensates for
the polyelectrolyte effect by masking the electrostatic repulsion of
cationic groups along the polymer backbone. This is shown in the lower
portion of Figure 6 where plots to two different types of viscosity
values10,11 for the polymer plus electrolyte show linear behavior.
IR spectroscopy is the most routine spectral characterization
technique available for polymers. The formation and IR characterization
of polymer thin films is facile,13 giving both qualitative14 and quantitative15
information. Figure 7 gives the spectrum of the oxazoline polymer.
Functional group identification can be required of the students,
although polymer spectra often display unexpected combination bands and
contaminant peaks from retained solvent and reactants.
 Return to
Macrolab Directory
Return to
Macrolab Directory






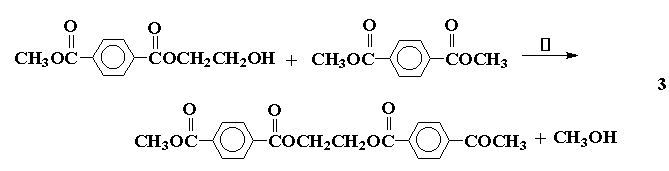


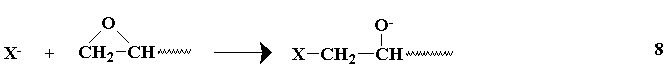


 =distance jaws moved (inches)
=distance jaws moved (inches)





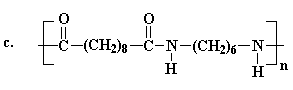
 Return to
USM Polymer Science Online Laboratory Directory
Return to
USM Polymer Science Online Laboratory Directory